
2015 Annual Report
31
Controlled
Drugs
Management
Medical
Devices
Management
Risk Assessment
Management and
Research Outcomes
Cosmetics
Management
Appendix
National
Laboratory and
Testing Network
Risk Communication
and Consumer
Protection
International
Cooperation and
Cross-Strait Exchange
Medicinal
Products
Management
Policy and
Organization
Food
Management
Section3 Medicinal Products Quality Chain Monitoring
Current Status
After a medicinal product is released to the market, there are still many uncertainties that may influence or
even impact product quality. These uncertainties may include changes to production processes as well
as transport and storage environments. Hence, TFDA is promoting the Good Distribution Practice (GDP)
for medicinal products to ensure the quality of product storage, transport, and distribution processes.
A comprehensive post-market quality chain monitoring system must be deployed, with measures such
as establishing a reporting system, active monitoring of global medicinal product quality alerts, using risk
assessments for selecting items, integrating the resources of local health bureaus, and implementing
quality monitoring of medicinal products sold on the market. In order to prevent the sales of counterfeit or
prohibited drugs through improper sales channels and other acts that seriously endanger public health
and safety, one of the key work items of health agencies must be the suppression of illegal drugs and illicit
advertisements.
Policies and Outcomes
1. Quality Monitoring of Medicinal Products
(1) Drug Product Defect Reporting System
Taiwan established a Reporting System of Medical Product Defects in 2004 to enable medical
personnel and the general public to immediately report any medical products with suspected
defects, allowing government agencies to take swift action. In 2014, a total of 845 medical product
defect reports were received, of which 150 were classified as high-risk defects. A total of 14 medical
product recalls was made as well.
(2) Quality Monitoring of Medical Products Released to the Market
Quality monitoring items have been selected in response to medicinal product management and policies,
risk assessment factors, and requirements of county- and city-level health departments in order to
safeguard medicinal product use quality and safety of the general public. In 2014, a total of 372 cases
was sampled and inspected of which 365 cases were found to be compliant while seven were found
to be nonconforming. The nonconforming products were officially handed over to the health bureaus
or departments in charge of the original inspections while the responsible manufacturers were notified
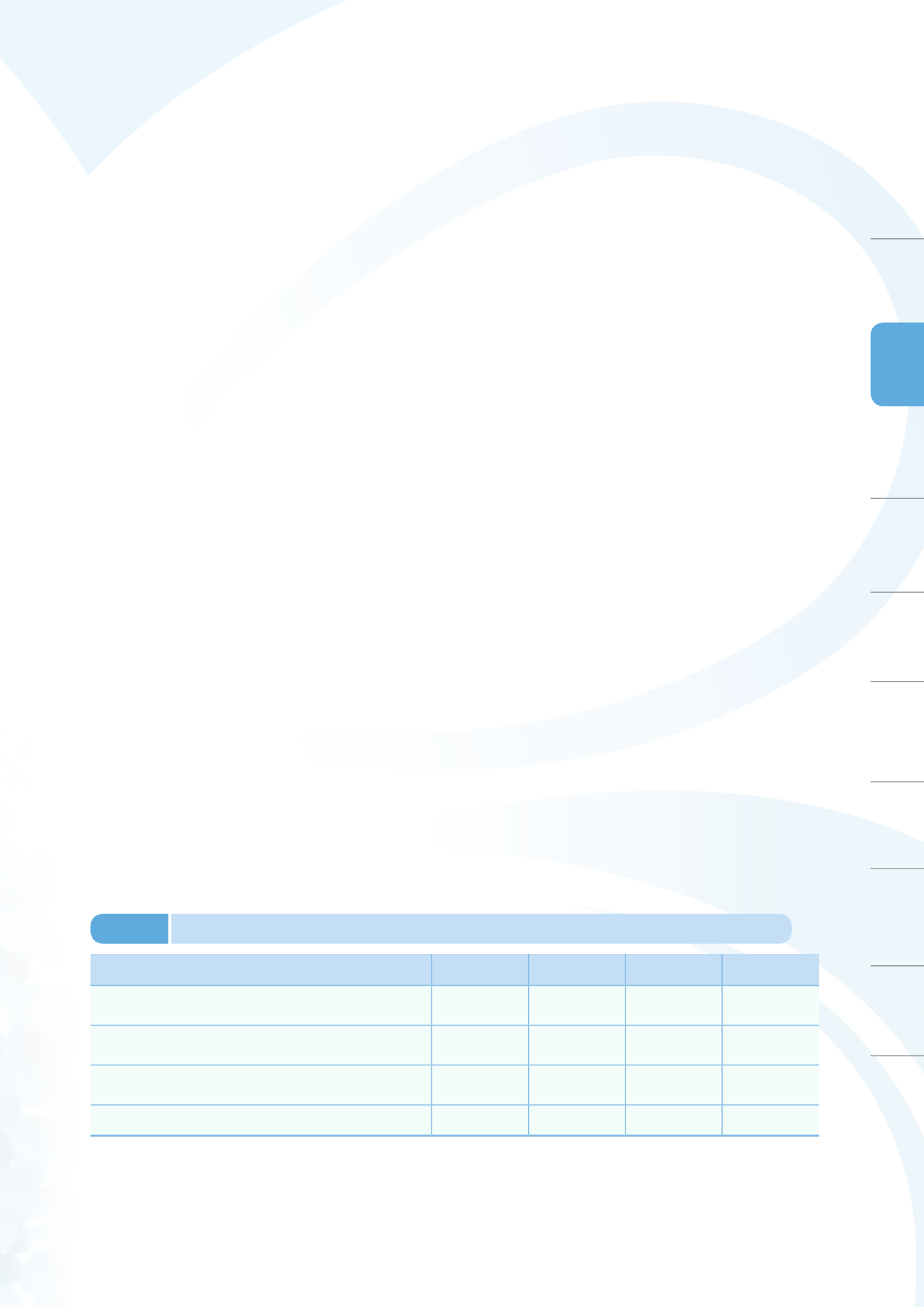
to conduct product recalls. Table 3-2 shows the monitoring results of the project. Detailed statistics on
quality monitoring of released medicinal products are presented in Appendix 1 Annex Table 9.
Project title
Total cases Conforming
cases
Nonconforming
cases
Conformity rate
(%)
Surveillance on the quality of antidiabetic, NSAIDs ,
hypnotic and antibiotic preparations in Taiwan
90
87
3
a
96.7
Post-market surveillance of varicella and MMR live
attenuated vaccines
148
148
0
100.0
Post-market quality surveillance and testing for foreign
materials in Traditional Chinese Medicine preparations
134
130
4
b
97.0
Total
372
365
7
98.1
Note: a. One case of dissolution test result nonconformity of anti-diabetic medication and two cases of dissolution test
result nonconformities for anti-inflammatory painkillers.
b. Two cases in nonconformities of total aerobic microbial counts and two cases of total ash.
Table 3-2
Medicinal Products quality monitoring and testing results in 2014



















