
53
Food and Drug Administration
Chapter
︱
Cosmetics Management
︱
5
Cosmetics have become essential goods in daily life. The stability, quality, and safety to
human health for long-term use have become public focus. The concerns on cosmetic
quality and safety have also induced the attention of the management of cosmetic
quality and safety in every country.
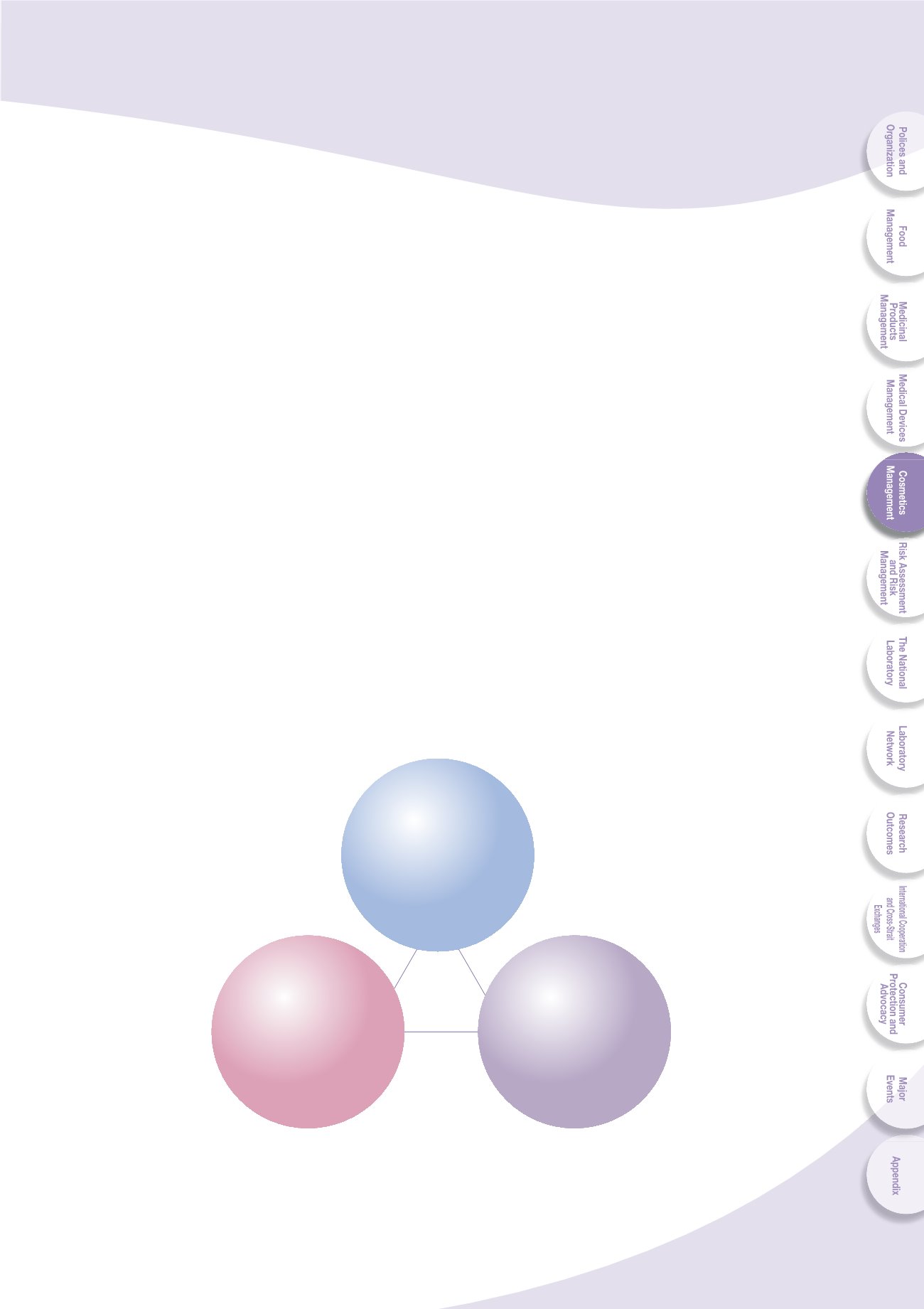
The current management infrastructure is divided into three elements: source control,
premarket management and post market surveillance (Fig. 5-1). The measure for
ZV\YJL JVU[YVS PZ [OL ]VS\U[HY` JVZTL[PJ .47 ;OL WYL THYRL[ THUHNLTLU[ PUJS\KLZ
the pre-market registration of medicated cosmetics and color additives as well as
the pre-market cosmetics advertisement review. The post-market surveillance is
implemented by the quality monitoring program and cross-county joint inspection.
Cosmetic product defect reporting system is built. Consumer advocacy for safe use of
cosmetics is also strengthened to construct a comprehensive cosmetics quality and
safety network.
Fig. 5-1 Current cosmetic management framework
Source Control
Cosmetics
Post-market Surveillance
Pre-market Management
Voluntary cosmetics
good manufacturing
practices
Registeration of
medicated cosmetics
Advertisement review
Quality monitoring and
joint insepection
Cosmetic product defect
reporting system
Security monitoring and
security advocacy



















