
27
Food and Drug Administration
Different from consumer goods, medicinal products are highly regulated. In order to
ensure public health, the regulations of medicinal products need to be harmonized
with international standards, as well as simplified and integrated in review process.
The source and distribution of medicinal products (including active pharmaceutical
ingredients) have to be regulated. Post-marketing surveillance is conducted to ensure
the quality and safety of medicinal products. In addition, investigation of illegal drugs
and controlled drug managements are taken to protect public safety.
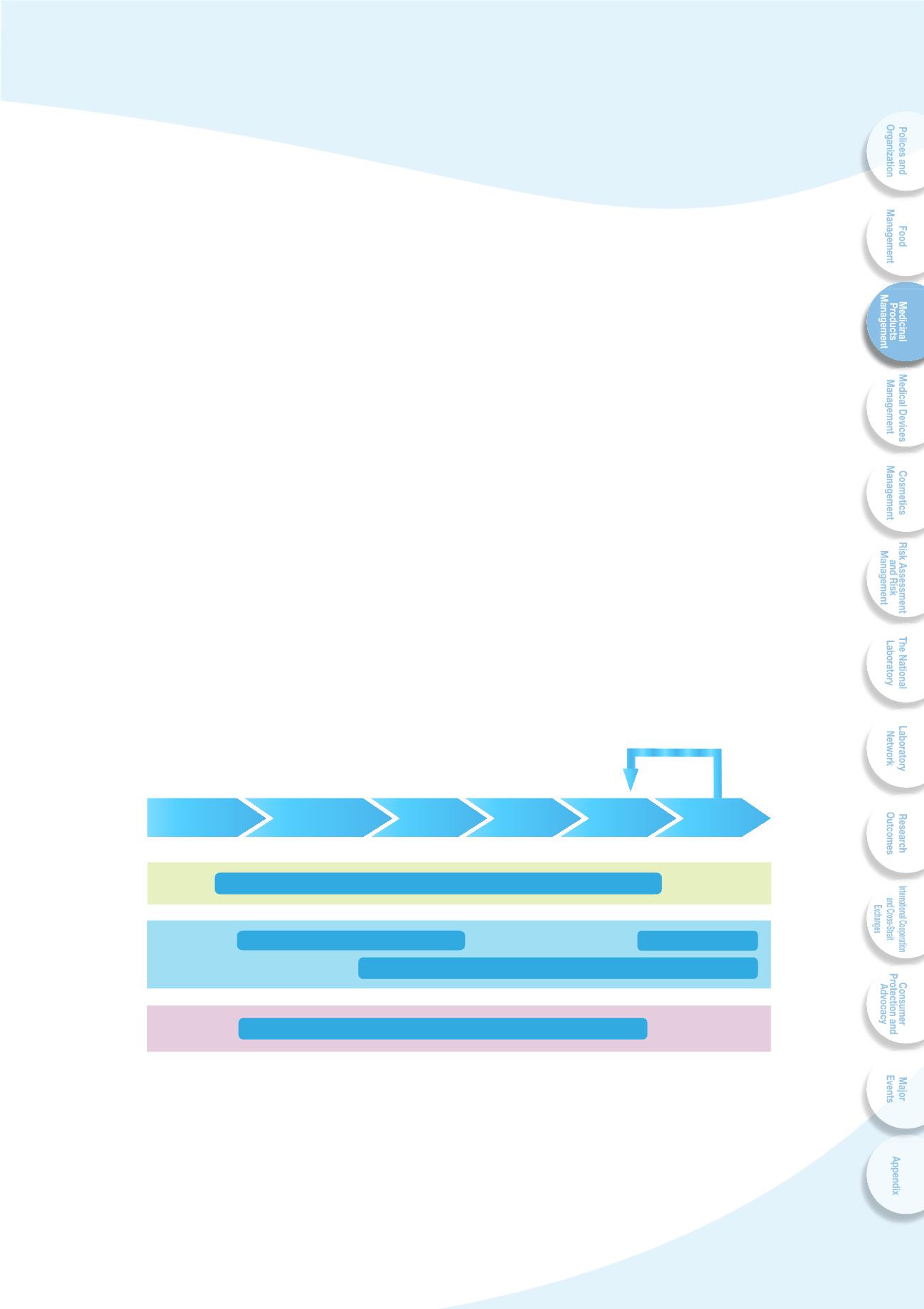
4VYLV]LY H WYVK\J[ SPMLJ`JSL THUHNLTLU[ Z`Z[LT PZ LZ[HISPZOLK PUJS\KPUN WYL
marketing approval and post-marketing surveillance system. The requirements of
pharmaceutical registration include development information, results of nonclinical
studies, clinical trials, stability tests, and chemical manufacture control documents with
common technical dossier. Inspections of GXP and post-marketing monitoring are
also executed to achieve comprehensive drug life cycle requirements (Fig. 3-1).
Fig. 3-1 Drug life cycle
CTD : Common Technical Document
G L P : Good Laboratory Practice
GCP : Good Clinical Practice
GPvP : Good Pharmacovigilance Practice
GMP : Good Manufacturing Practice
Review
Site
Inspection
Assay
Basic
research
Nonclinical
study
Clinical
trial
Registration
Manu-
facture
Marketing
CTD: safety, efficacy, quality
GLP
、
GCP
GPvP
GMP
assay/analysis
Chapter
︱
Medicinal Products Management
︱
3



















