
27
Food and Drug Administration
ୋ
z
ᖹۜ၍ଣ
z
3
ྡ
3-1
c
ᖹۜ͛նಂ
ᖹۜၾɓছऊ൬ۜʔΝdҹᗫɛ͏Ԓੰʿ͛նτΌd᙮৷
၍ܓ
Փପุd̀
՟ʕ̯ሊ͛˴၍ዚᗫࣨ೯ʘᖹ̙ۜᗇʑঐɪ̹னਯdᖹۜ
ج
ࠈࡌ
ၾყટ
ࠐ
eᄲ
ݟ
ᔊʷၾ၍ଣɓʩʷe๕᎘Ⴁிၾ၍ଣe
ݴ
ஷၾۜሯ္eʔ
ج
ᖹ
՟ي
ᇕʿ၍
Փᖹۜ၍ଣdᎇࣛછԨ੶ʷᖹۜτΌ္ዚՓdᆽ
ڭ
͏͜ᖹτΌf
ܔ
ͭҁᖹۜ͛նಂdܼ̍ɪ̹
ۃ
ʿɪ̹
ܝ
ʘ၍ଣdႊᖹۜɪ̹
ۃ
ਿᓾ
Ӻe
ڢ
ᑗґ༊᜕eᑗґ༊᜕eᎴԄႡிච
ݬ
˸ʿɪ̹
ܝ
Դ͜eΫ㉿ʿܵᚃҷഛഃච
ݬ
f
މ
ᆽ
ڭ
ᖹۜۜሯʿᐕࣖd˸ɪ̹
ݟۃ
᜕೮াᄲ
ݟ
ஷ͜Ҧஔ˖eᖹۜᅵۜᏨ᜕e
ධᖹۜᎴԄᇍ
GXP
ഃᇆ
ݟ
Փ
ܓ
d˸ʿɪ̹
ܝ
ʘᖹۜۜሯτΌ္ԣᚐၣЪ
ุd˸༺Όࠦᖹۜ͛նಂ
ࠅ
Ӌʘͦᅺྡ
3-1
f
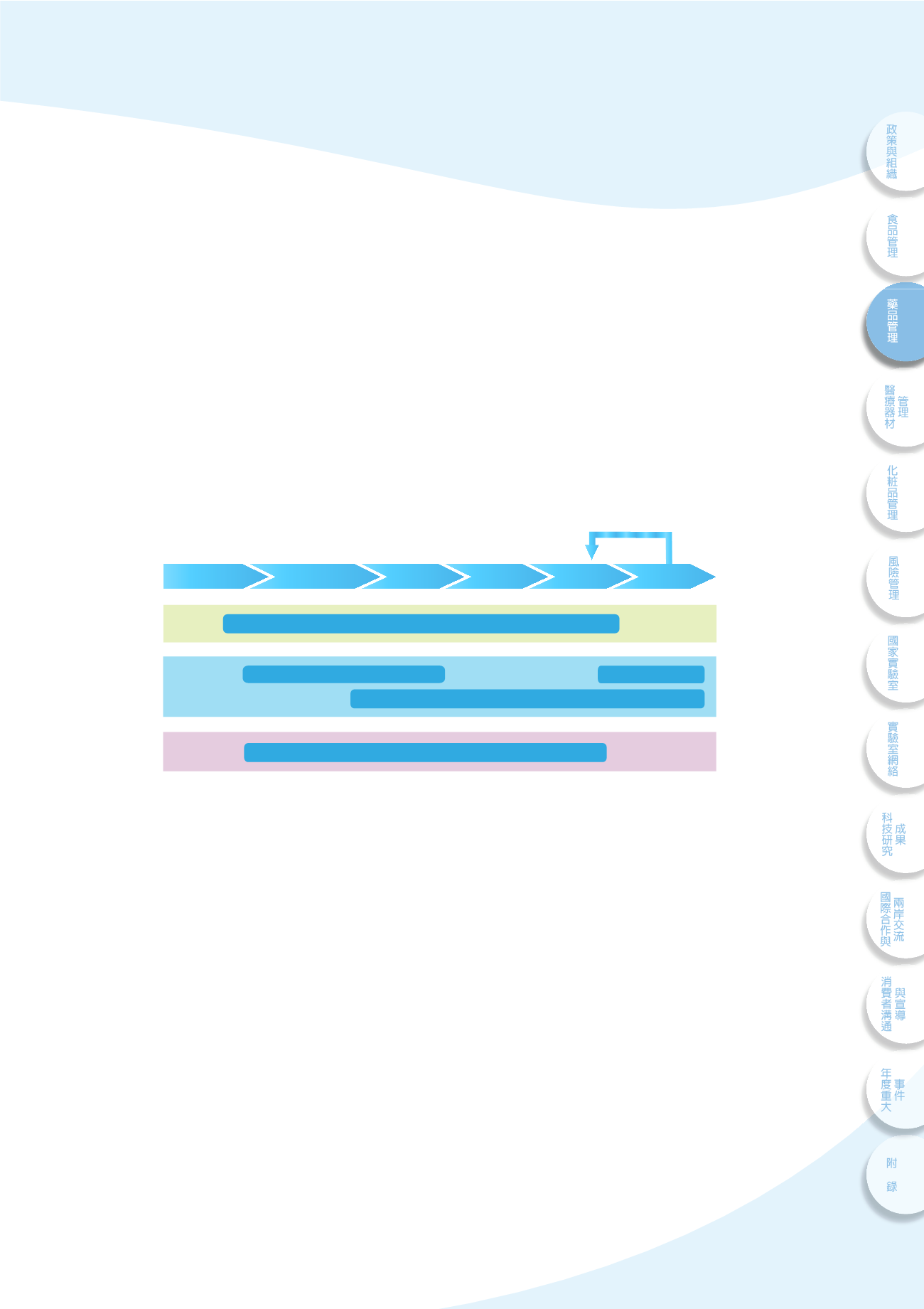
ୋɓືcᖹۜ
ج
ᅺʿପۜᄲ
ݟ
ତ
ر
ყ
ج
࿁ɪ̹
ܝ
ᖹۜʘۜሯ
ࠅ
Ӌϋ৷dˈՉ݊ɪ̹
ܝ
ᜊһ೮াʘ၍ଣdʿ
ࠦ࿁อጳۨ࿒ᖹۜᚃɪ̹Էνj
ߤ
eਿΪ
ط
ᐕପۜdϾlzᗫ
ج
ᕍνd
ΪϤ͉ܵᚃᏨൖ
ࡌ
͍ʿ
ࠈ
ᖹ
ي
ᗫ၍ଣᇍd̤ਞϽყගᖹۜ၍ଣᒈැʿ০࿁
ࠬᎈ༰৷e͜࿁Ոतମʘᖹۜ၍ଣd
ࠈ
֛ᖹۜ
ݟ
᜕೮া၍ଣՓ
ܓ
Ԩᚃᄣ
ࡌ
ᖹ
݁၍ଣ
ج
˸ᆽ
ڭ
ᖹۜۜሯɓ
ߧ
ၾϞࣖf
CTD
GLP
GCP
GPvP
GMP
j
Common Technical Document (
ஷ͜Ҧஔ˖
)
j
Good Laboratory Practice (
ᎴԄྼ᜕܃Ъᇍ
)
j
Good Clinical Practice (
ᖹۜᎴԄᑗґ༊᜕
ۆ
)
j
Good Pharmacovigilance Practice (
ᖹۜᎴԄτΌ္ൖᇍ
)
j
Good Manufacturing Practice (
ᎴԄᖹۜႡிᇍ
)
ᄲ
ݟ
ᇆ
ݟ
Ꮸ᜕
ਿᓾӺ
ڢ
ᑗґ༊᜕ ᑗґ༊᜕ ͡ሗ̙ ͛ପႡி ɪ̹
ݴ
ஷ
CTD
τΌeϞࣖeۜሯ
GLP
e
GCP
GPvP
GMP
Ꮸ᜕ ʱؓ



















