
33
Food and Drug Administration
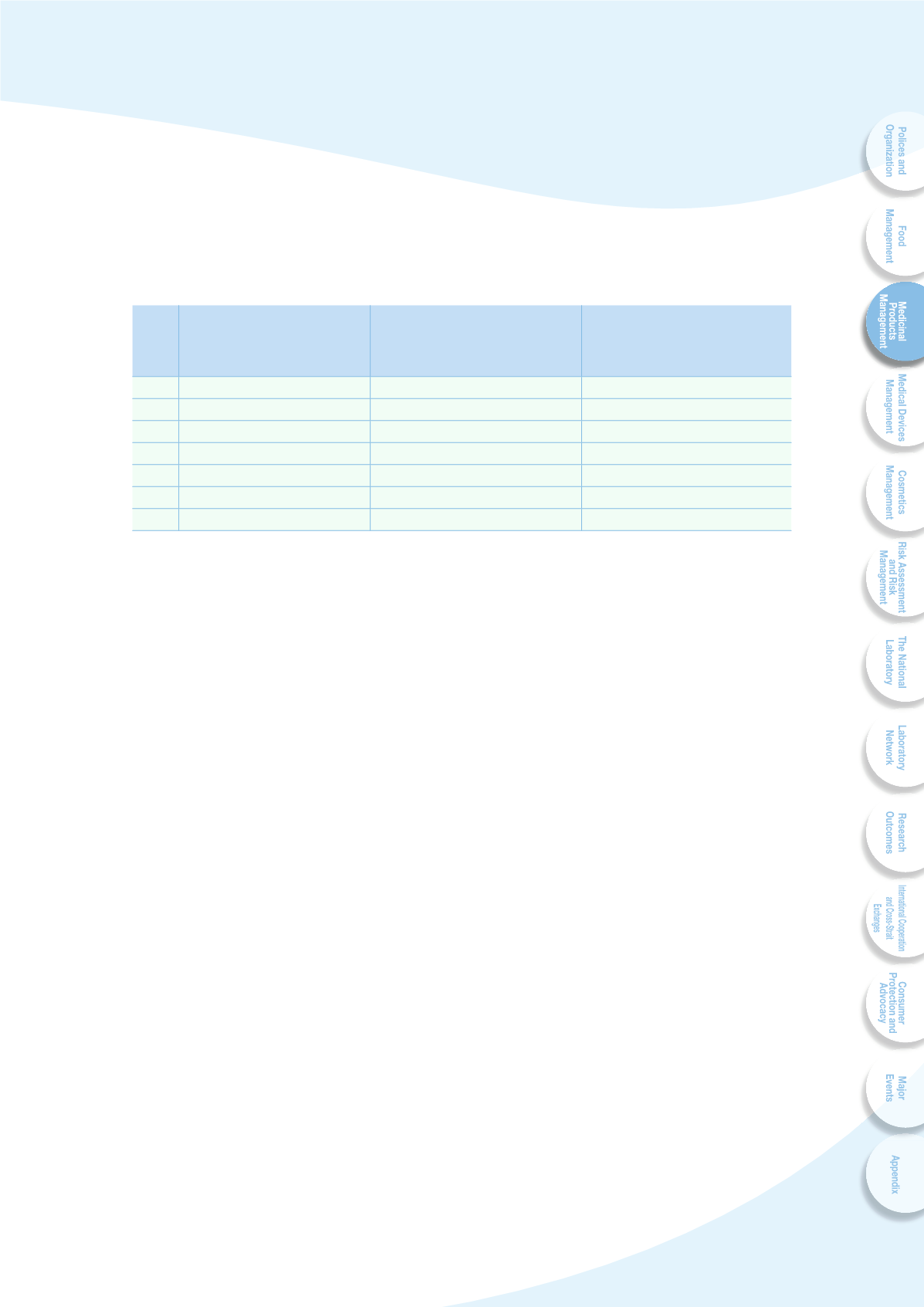
Table 3-2 Status of GMP compliance of domestic and foreign pharmaceutical
manufacturers
Year
No. of domestic
western pharmaceutical
manufacturers compliant
^P[O .47
No. of domestic western
pharmaceutical manufacturers
JVTWSPHU[ ^P[O 70* : .47
No. of foreign western
pharmaceutical manufacturers
JVTWSPHU[ ^P[O .47 HM[LY VU
site inspection
2007
160
-
93
2008
151
-
118
2009
154
5
140
2010
155
22
157
2011
149
33
180
2012
145
44
209
2013
140
57
213
J ( [V[HS VM
KVTLZ[PJ WOHYTHJL\[PJHS THU\MHJ[\YLYZ OHK JVTWSPLK ^P[O 70* : .47
Guide by the end of December 2013. This positive impact promotes the cooperation
among international pharmaceutical trading business and contact manufacturing.
The export of the medicinal products had increased double in recent four years.
;OL PTWSLTLU[H[PVU VM 70* : .47 MVY [OL TLKPJPUHS NHZLZ THU\MHJ[\YLYZ
a. TFDA had helped the medicinal gases manufacturers to meet the requirements of
70* : .47 .\PKL ZPUJL
;OL ZJVWL JV]LYLK [OL MPLSKZ VM TH[LYPHSZ Z\WWS`
production control, quality control, quality management system as well as the
prevention of cross contamination to assure the quality of medicinal gases.
b. A total of 28 medicinal gases manufacturers had met the requirements of PIC/S
.47 .\PKL VU +LJLTILY
[OL K\L KH[L VM [OL PTWSLTLU[H[PVU VM 70* :
.47 VU TLKPJPUHS NHZLZ THU\MHJ[\YLYZ
2. Product Sources Management for Pharmaceutical Manufacturers
(1)
“
+Y\N 4HZ[LY -PSL +4-
”
polity was established to fortify the active pharmaceutical
ingredients (API) management. From October 2009 to the end of 2013, the total
U\TILY VM +4- HWWSPJH[PVUZ ^LYL
"
VM [OLT ^LYL HWWYV]LK
approved).
;-+( OHK HKVW[LK 70* : .47 .\PKL MVY HJ[P]L WOHYTHJL\[PJHS PUNYLKPLU[Z
Furthermore, on September 25, 2013, TFDA had announced API manufacturers
ZOV\SK JVTWS` ^P[O 70* : .47 .\PKL I` +LJLTILY
)` [OL LUK VM
+LJLTILY
[OLYL ^LYL (70 P[LTZ JVTWSPHU[ ^P[O 70* : .47 .\PKL ^OPJO
were manufactured by 21 API manufacturers.



















